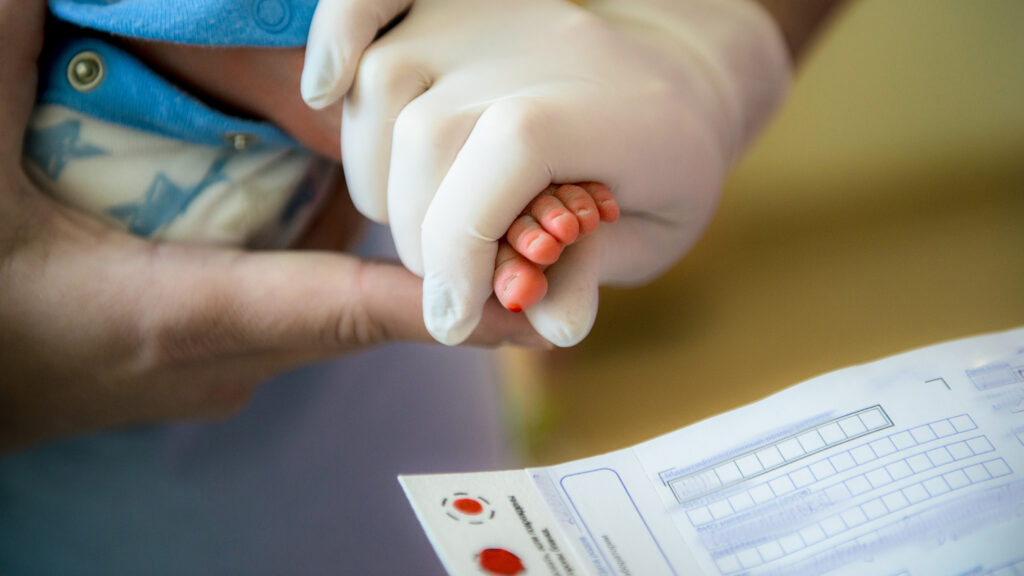
TYears ago, clinicians at a handful of hospitals in the United States began sequencing the genomes of apparently healthy babies to understand how the technology could reveal hidden genetic conditions that are undetected by routine newborn blood tests. New research from one such study suggests that the impact of having that kind of information extends far beyond the baby whose DNA is being decoded.
In a study Published Monday in the American Journal of Human Genetics, researchers from Mass General Brigham and Boston Children’s Hospital reported that of the first 159 babies screened by genomic sequencing, 17 were discovered to have unexpected mutations in disease-related genes.
Over the next three to five years, in the majority of the 17 babies’ families, these discoveries led parents and other relatives to undergo additional tests that led to uncovering the cause of illnesses running through their family tree . In three cases, mothers who learned they were carriers of a gene that dramatically increased their risks of certain cancers opted to have prophylactic surgeries to reduce those risks — a finding the lead researcher says undermines ethical objections to informing women. families about genetic findings, even if they are not. t immediately usable for the newborn.
“This is a real-world refutation of the prevailing view that we shouldn’t share variants with disease risks in adulthood in children,” said Robert Green, a medical geneticist at Harvard and Brigham and Women’s Hospital who is leading the BabySeq study that produced the new research. “There are ethicists who say that a child should not be used as a genetic canary in a coal mine – that one member of a family should not be used as an entry point for an entire family without their consent, but I want That. Look at these mothers. We probably saved their lives. Are you really going to compare that to a theoretical loss of autonomy at some point in the child’s future?
As the cost of DNA sequencing falls, the prospect of screening the whole genome of millions of newborns has raised major concerns about how useful that information really is. Experts are divided on whether the benefits of early disease detection outweigh the additional costs and burden on the health care system, as well as the potential psychological impact on families of knowing they carry disease risk genes and on the child if that decision is made before them before they are old enough to walk, talk and consent.
Other clinical trials are underway evaluating the health benefits, financial costs and ethical implications of sequencing compared to the standard blood tests all newborns receive to identify a limited number of inherited conditions. They include a number federally funded studies in the United States, as well as a pilot program in the United Kingdom that will sequence the genomes of 100,000 newborns over the next two years. They each give different amounts of genetic information back to families and their doctors. But only BabySeq offers a glimpse into what’s lurking in 78 genes linked to an increased risk of diseases that develop in adulthood.
BabySeq is a clinical trial involving several hundred families, some with sick babies, some with healthy ones; half of the children received standard neonatal screening and half received screening plus sequencing. When the trial first launched in 2013, Green and his colleagues went back and forth on how much genetic information to give back to parents about their new child. Initially, they decided to only disclose genetic variants involved in a range of childhood-onset disorders. But around that time, two major things happened that changed their mind.
The first was that the American College of Medical Genetics and Genomics issued a new recommendation that the incidental finding of one of 56 genes for “highly useful” conditions – that is, things you could diagnose and treat, or if you not treat, at least control — are reported to all subjects undergoing clinical genomic sequencing, regardless of age. Three of those conditions did not appear until adulthood: cancer caused by variants in the BRCA genes and an aggressive form of colon cancer, Lynch syndrome.
The second was what happened to one of the first participants to join BabySeq’s sequencing arm. The baby boy was born with a serious heart problem and soon died. But lurking in his DNA data, the finding of a BRCA variant was associated with a 45% increased risk of breast cancer in women, as well as an increased risk of other cancers in both sexes. The BabySeq team was able to deduce from saliva samples from the parents that the mutation came from the mother. But because of the research protocol, they weren’t allowed to tell the family what they found.
The awkward situation caused them to rethink their strategy and rework their protocol. As a result, all subsequent families who participated in BabySeq were given the opportunity to receive information about genes that cause disease risk in adulthood. According to the latest data from the survey, most families chose it. In 13 of the 17 babies discovered to have disease-related mutations, the information they received led to additional screening for family members at risk. The experience has had a major impact on how the BabySeq project is now expanding.
Green and his collaborators recently began recruiting for a second phase of the trial, which aims to enroll more than 1,000 infants and their families from racially, ethnically and socioeconomically diverse communities in Boston, New York and Birmingham, Ala. The first BabySeq study overwhelmingly wealthier, college-educated people with European ancestry, making findings not very generalizable to the wider U.S. population. Other changes in this phase include recruiting slightly older babies – up to 6 months old.
Parents who opt in to this phase are informed that they will receive genetic information about their child related to conditions that develop in adulthood, but there is no way to opt out without refusing participation altogether.
“What this whole thing helps us think about is different ways we think of benefits for families,” Green said. “I think it’s a good thing to find these variants in babies for the benefit of the whole family.”